The atomic radius for atoms of an element tends to go up as you move down a group of elements in the table. While the atomic radius can be defined in a number of different ways, the general atomic radius trend across the periodic table holds true. Van der Waals radius refers to the distance that exists between the closest approach of two non-bonded atoms in a defined element. The metallic radius is usually defined as being half of the distance between two metal ions adjacent to one another in a metallic lattice. The metallic radius refers to a specific type of chemical bonding between positively charged metal ions and conduction electrons. The covalent radius is the size of an atom that makes up a portion of a covalent bond. Ionic radius refers to the measure of an atom’s ion when in a crystal lattice, and it’s typically half the distance between the nuclei of two different ions which are barely touching. “I must confess I am jealous of the term atom for though it is very easy to talk of atoms, it is very difficult to form a clear idea of their nature.” - Michael Faraday Various methods of measuring atomic radius are used, including the ionic radius, the metallic radius, the covalent radius, and the Van der Waals radius. First of all, it’s important to be aware of the fact that although the term atomic radius is used to refer to an atom’s size, there isn’t an agreed-upon definition for this value.

Yet this value isn’t as easy to pin down as you might think. What Is Atomic Radius?Ītomic radius refers to the size of the atom.
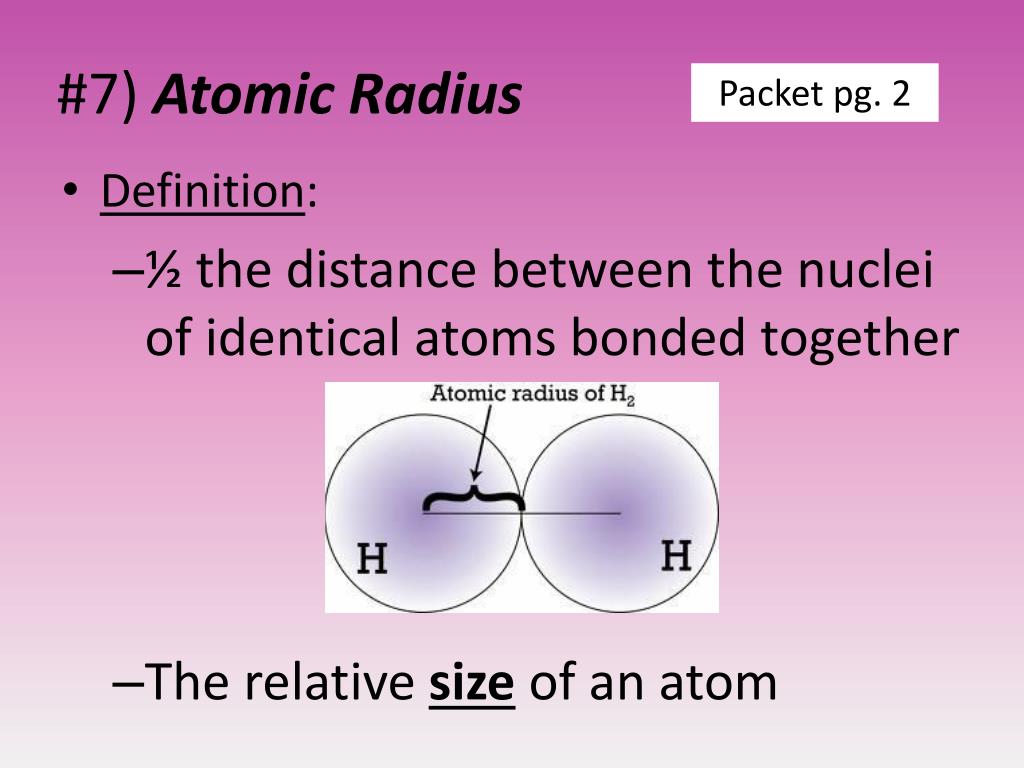
To understand why this happens it would be helpful to take a close look at the definition of atomic radius and the radius of different elements in the periodic table. In general, the atomic radius of an element tends to increase as you move down an element group in the periodic table. The atomic radius trend describes how the atomic radius changes as you move across the periodic table of the elements.
